Richards Group
Current and past research
Shape dependence of phagocytosis
The effect of target shape on engulfment during phagocytosis has applications to bacterial infection, immunotherapy and the design of microparticle drug delivery systems. In collaboration with Dr Peter Petrov and Dr Charlie Jeynes, we study the effect of shape on the rate of neutrophil and macrophage phagocytosis. This involves dual-micropipette experiments (see image) that allows fine control over cell-target interactions and gives high-resolution images of the shape of the phagocytic cup during the whole engulfment process.
The multistage nature of phagocytic engulfment
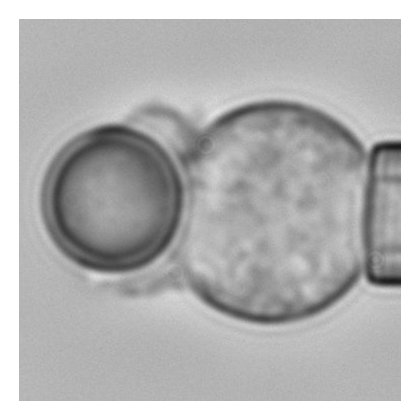
Using a combination of mathematical modelling, computer simulations and image analysis, this work showed that engulfment during phagocytosis proceeds in two distinct stages, an initial slow stage followed by a much quicker second stage.
Papers:
Computer simulations of early embryogenesis
The very earliest stages of mammalian embryogenesis, from the single fertilised egg to the mature blastocyst, sets the basis for subsequent development. Understanding this process has important applications to human fertility, IVF treatment (where most embryos fail before implantation), conservation of endangered species and food security (via animals such as pigs, cows and sheep). In this work, we use a variety of modelling frameworks to simulate the first few days of development, focusing on the role of biophysical forces such as membrane tension and cell-to-cell adhesion.
Recent review: Cockerell A, Wright L, Dattani A, Guo G, Smith A, Tsaneva-Atanasova K, Richards DM: Biophysical models of early mammalian embryogenesis. Stem Cell Rep 18:26 (Stem Cell Reports)
Automatic image analysis of muscle ultrasound

Muscle quality can become greatly reduced due to injury or long periods of rehabilitation. Ways of quantifying muscle quality is therefore important to determine the progress and success of treatments such as physiotherapy. Ultrasound imaging is a potentially affordable way to do this. We are developing image analysis techniques to automatically extract information on muscle quality in realtime from ultrasound images, with the aim of improving clinical practice and patient treatment in the rehabilitation clinic.
Machine learning of microglial state
Microglia are the macrophgaes of the brain, dedicated immune cells that travel around searching for unwanted objects. Chronic activation of microglia is a driver of neurodegenerative diseases such as Alzheimer’s and Motor Neuron Disease. Microglia can exist in different states, often characterised by markedly different cell morphology and dynamics. We are trying to use machine learning, including deep neural networks, to automatically classify microglial state based on cell shape.
Understanding the growth of filamentous fungi

The fungal pathogens, C. albicans and A. fumigatus, cause over 600,000 infections a year worldwide, with death rates up to 95%. Both fungi form long tubes, called hyphae, that spread through the body causing inflammation and organ failure. This work aims to understand how hyphae form and how they invade tissue. This involves a combination of mathematical modelling, computer simulation and automatic image analysis. The long-term aim is to predict new ways that fungal infections might be stopped, potentially leading to novel types of drug that will allow treatment of two of the world's most deadly fungal diseases.
Analysis and modelling of calcium signalling in fungi
Calcium is an important second messenger in eukaryotic cells, but is poorly understood in fungi. The little work that has been performed on calcium in fungi has focussed almost exclusively on Saccharomyces cerevisiae. Various fungal cells exhibit intracellular calcium spiking. This work aims to understand this and determine if such spiking can be described by a Hodgkin-Huxley-type model. In particular, we study the frequency, amplitude and shape of fungal calcium spikes, including how these are affected by various drug treatments.
Phagosome dynamics
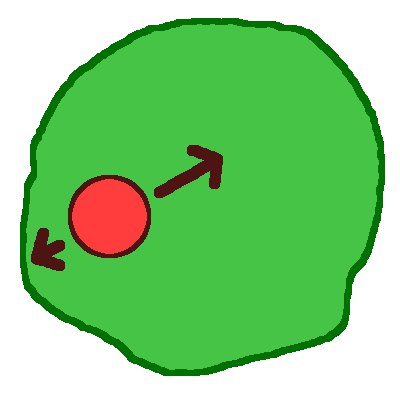
Once phagocytic engulfment is complete, the target is completely within the cell, inside a membrane-bound compartment called the phagosome. The phagosome moves towards the centre of the cell via a mixture of inward motion, random jiggling and stationary stages. This project involves a collaboration with Professor Holger Kress at Universität Bayreuth in Germany, who provides the experimental data. We are developing a theoretical model (see image) for phagosome motion towards the perinuclear region, including a combination of diffusion, active motion and stalling.
Dynamics of peroxisome shape

Peroxisomes are tiny, specialised organelles within cells that are involved in various reactions such as the breakdown of fatty acids. When necessary, they can elongate and divide in order to produce new peroxisomes. In collaboration with Dr Michael Schrader in Biosciences at Exeter University, this work studies the dynamics of peroxisomal membrane shape (see image), including which forces account for peroxisome extensions during organelle division. The aim is to not only understand the cell biology involved, but to also examine various diseases related to defective peroxisomes such as adrenoleukodystrophy (ALD).
Papers:
- Castro IG, Richards DM, Metz J, Costello JL, Passmore JB, Schrader TA, Gouveia A, Ribeiro D, Schrader M: A role for MIRO1 in motility and membrane dynamics of peroxisomes. Traffic 19:229-42 (PubMed)
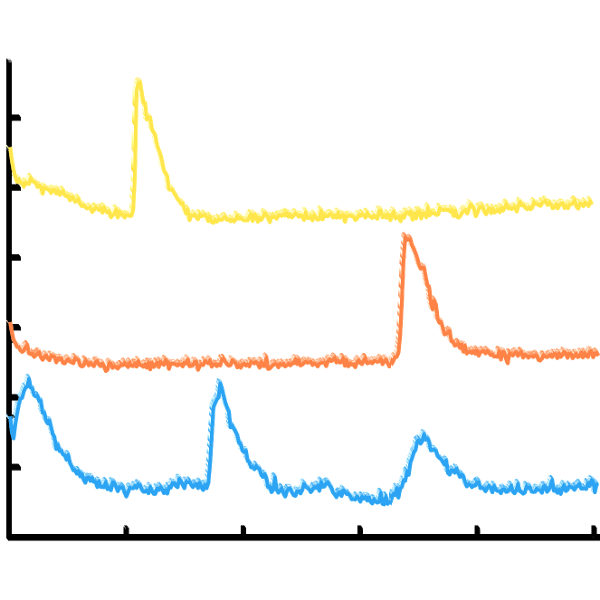
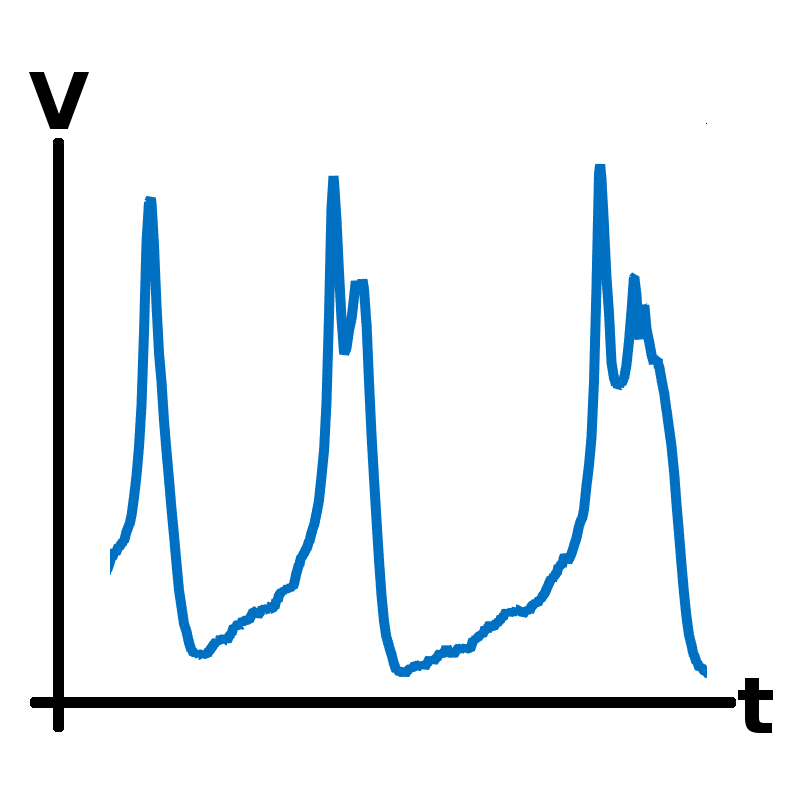
The role of noise in pituitary cells
Endocrine cells in the pituitary gland secrete hormones that regulate essential functions such as growth, metabolism and stress. Hormone secretion is controlled by regulatory signals from the brain that act on ion channels in the cell membrane, affecting the cell’s electrical activity. The ion channels are stochastic elements that randomly fluctuate between open and closed states. This project, involving a collaboration with Dr Joël Tabak and Dr Jamie Walker at Exeter University, uses mathematics (see image) to study the role of channel stochasticity in regulating the electrical activity of these cells.
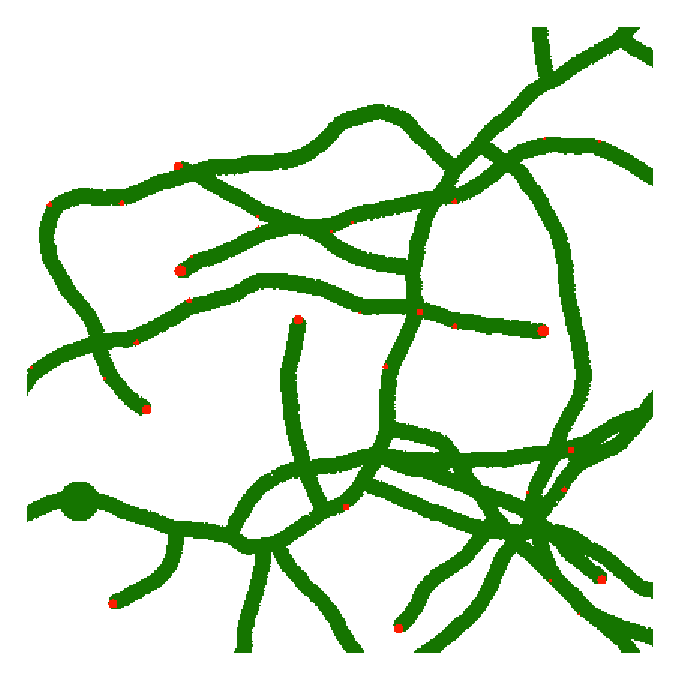
Plant response to phytopathogens

Plants are continually attacked by pathogens such as certain fungi. They respond by local delivery of membrane-integrated proteins to the site of infection, causing modification of the cell wall and the membrane that are crucial for host defence. However, how these proteins are transported to the site of infection is, at present, almost completely unknown. Based on a collaboration with Dr Mike Deeks in Biosciences at the University of Exeter, this project uses computer simulations (see image) to examine the plant immune response to phytopathogens. The overall future aim is to begin to understand how pathogens exploit weaknesses to cause disease and identify strategies to reinforce the plant immune system.
Branching in Streptomyces
Modelling of branching in the filamentous bacterium Streptomyces, where a critical role is played by the protein DivIVA. This work showed that new foci of DivIVA arise from an entirely novel mechanism, with new foci breaking off from existing tip foci.
Papers:
- Richards DM, Hempel AM, Flärdh K, Buttner MJ, Howard M: Mechanistic basis of branch-site selection in filamentous bacteria. PLOS Comput Biol 8(3):e1002423 (PubMed)
- Hempel AM, Cantlay S, Molle V, Wang SB, Naldrett MJ, Parker JL, Richards DM, Jung YG, Buttner MJ, Flärdh K: The Ser/Thr protein kinase AfsK regulates polar growth and hyphal branching in the filamentous bacteria Streptomyces. Proc Natl Acad Sci USA 109(35):E2371-9 (PubMed)
- Flärdh K, Richards DM, Hempel AM, Howard M, Buttner MJ: Regulation of apical growth and hyphal branching in Streptomyces. Curr Opin Microbiol 15(6):737-43 (PubMed)
Telomere bouquet formation during meiosis
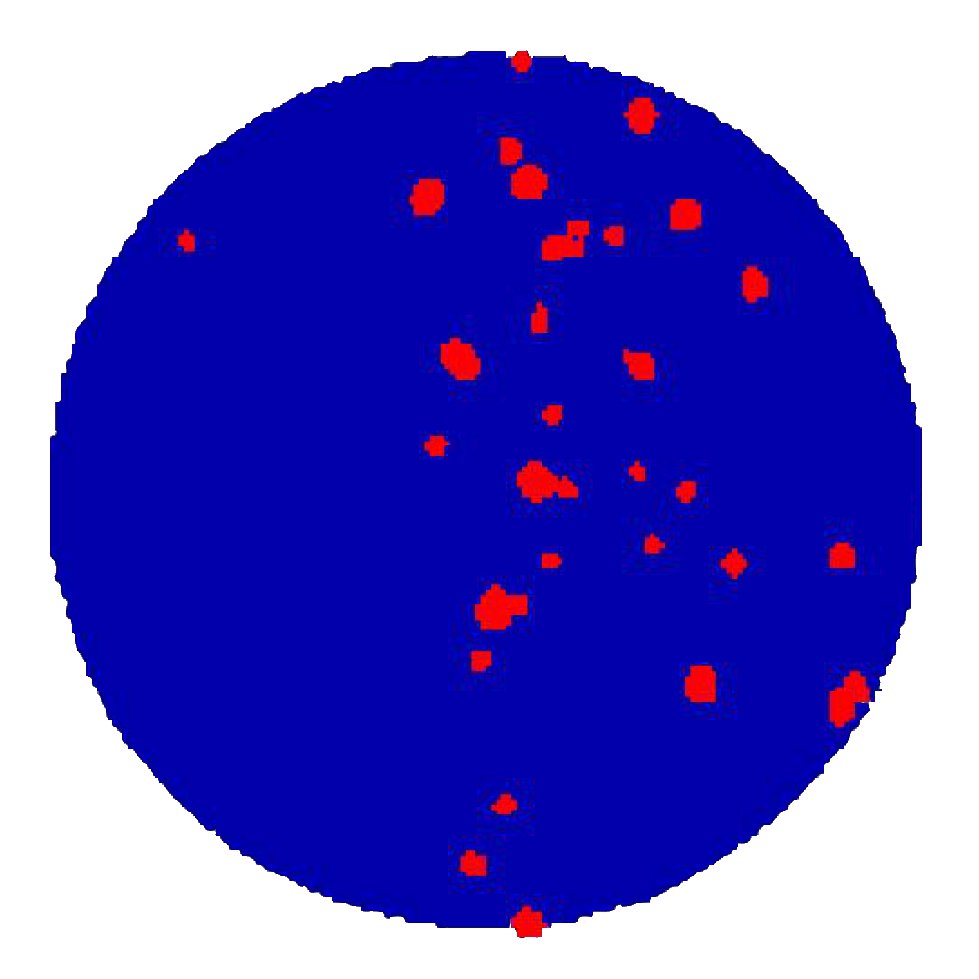
This work modelled how homologous chromosomes pair during meiosis, confirming the need for directed motion. For the first time we showed, using a combination of theoretical modelling and image analysis, that purely directed motion is sufficient to explain telomere nuclear motion.
Papers:
- Richards DM, Greer E, Martin AC, Moore G, Shaw PJ, Howard M: Quantitative dynamics of telomere bouquet formation. PLOS Comput Biol 8(12):e1002812 (PubMed)
- Richards DM, Berry S, Howard M: Illustrations of mathematical modelling in biology: epigenetics, meiosis, and an outlook. Cold Spring Harb Sym 77:175-81 (PubMed)
Derivatives in morphogenesis
This work studied the robustness of boundary specification when readout involves morphogen concentration derivatives, rather than the concentration itself, showing that such models can still lead to precise positioning.
Papers:
- Richards DM, Saunders TE: Spatiotemporal analysis of different mechanisms for interpreting morphogen gradients. Biophys J 108:2061-73 (PubMed)
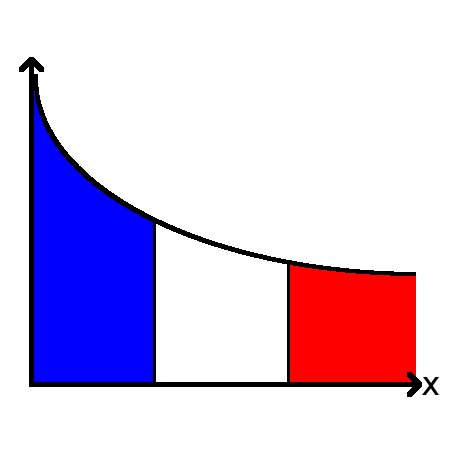
Accuracy in cell sensing
Many cells sense and respond to external concentrations, and there are various possibilities for how these concentrations can be encoded within the cell. This work studied the theoretical limits of these different possibilities.
Papers:
- Micali G, Aquino G, Richards DM, Endres RG (2015) Accurate encoding and decoding by single cells: amplitude versus frequency modulation. PLOS Comput Biol 11(6):e1004222 (PubMed)